Lithium battery cells have become essential in modern technology. Their applications range from smartphones to electric vehicles. Dr. Angela Yu, a leading expert in battery technology, once stated, "Lithium battery cells transform energy storage and redefine our power needs." This quote encapsulates the significance of these cells in today’s world.
At their core, lithium battery cells operate through electrochemical reactions. They store energy in lithium ions, enabling high efficiency and long-lasting performance. However, challenges remain. For instance, issues regarding safety and longevity must be addressed. While advancements continue, the need for sustainability is urgent.
Despite their advantages, lithium battery cells are not without flaws. For instance, the environmental impact of lithium extraction raises concerns. The industry must navigate these complexities while innovating. Understanding how these cells work allows for informed discussions on their future. Exploring potential improvements can unlock even greater benefits for users.

Lithium battery cells are essential components in modern energy storage systems. These cells provide power for various applications, from electric vehicles to portable electronics. A lithium battery cell typically contains an anode, a cathode, and an electrolyte. The anode is often made of graphite, while the cathode is composed of lithium metal oxide. When the battery discharges, lithium ions move from the anode to the cathode, generating an electric current.
According to a report from the International Energy Agency (IEA), the global demand for lithium-ion batteries is set to increase significantly, with an expected growth rate of 20% annually through 2030. This surge is largely due to the rising need for renewable energy solutions. Battery cells can store vast amounts of energy; for instance, a typical lithium-ion cell has an energy density of 150-250 Wh/kg. Yet, challenges remain, such as the environmental impact associated with lithium extraction and battery recycling.
Developments in battery technology continuously aim to enhance performance and sustainability. Innovative approaches are being explored, including solid-state batteries, which promise higher energy densities. However, transitioning to these technologies poses technical and economic challenges. The industry must address concerns over sourcing materials and ensuring long-term sustainability.
| Parameter | Description | Typical Value |
|---|---|---|
| Cell Type | Common types of lithium battery cells | Li-ion, LiPo, LFP |
| Nominal Voltage | Typical operating voltage of a single cell | 3.7V |
| Capacity | Amount of charge stored | 1500 - 3500 mAh |
| Cycle Life | Number of charge cycles before capacity significantly drops | 300 - 500 cycles |
| Energy Density | Amount of energy stored per unit mass | 150 - 250 Wh/kg |
| Temperature Range | Operating temperature limits | -20°C to 60°C |
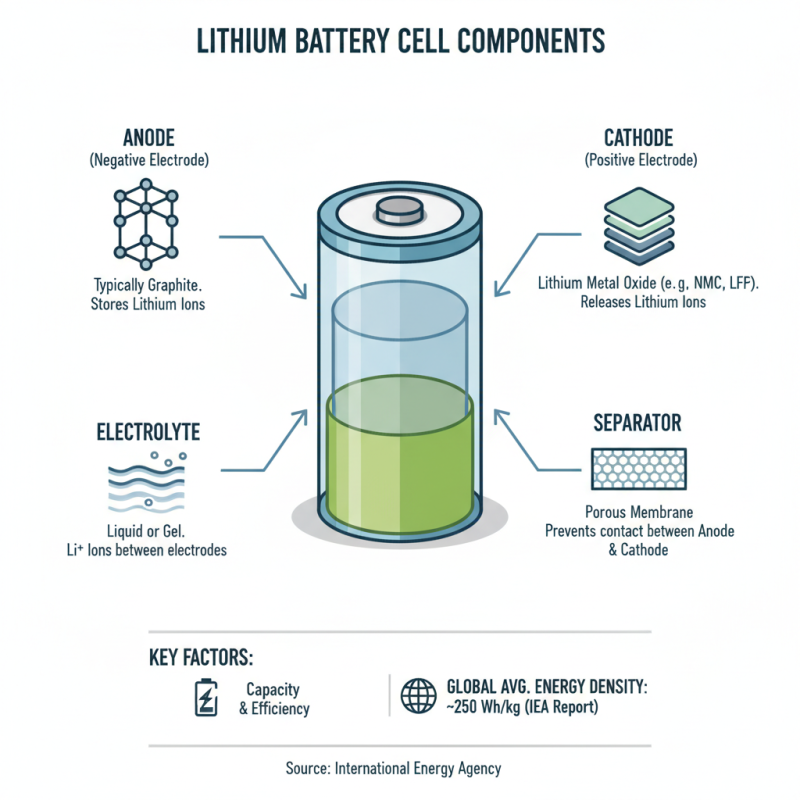
Lithium battery cells are fundamental components of modern energy storage systems. They consist of several key parts: anode, cathode, electrolyte, and separator. The anode usually contains graphite, while the cathode is often made of lithium metal oxide. These materials help determine the battery's capacity and efficiency. According to a recent report by the International Energy Agency, the global average energy density of lithium-ion cells is around 250 Wh/kg. This value can vary based on the materials used.
The electrolyte plays a crucial role in facilitating ion movement between the anode and cathode. Often comprised of lithium salts dissolved in a solvent, the electrolyte's composition significantly influences the battery's performance. Separators are vital for preventing short circuits by keeping the anode and cathode apart. They typically use porous materials to allow lithium ions to pass through while blocking electrons. This dynamic interplay of components ensures that lithium batteries deliver high energy output efficiently.
Tips: Always check the battery's specifications for optimal usage. Different applications may require specific battery types. Remember that battery lifespan can be affected by temperature and charging habits, so careful usage is essential. Regular maintenance can enhance performance and longevity, minimizing the need for replacements.
Lithium batteries are increasingly popular due to their efficiency and long life. The electrochemical process within these batteries is fascinating and complex. At the core, lithium ions move between the anode and cathode during charge and discharge cycles. This movement enables energy storage and release, powering countless devices we use daily.
In a typical lithium battery, the anode often consists of graphite, while the cathode is made from materials like lithium cobalt oxide. When a battery is charged, lithium ions are extracted from the cathode and move through the electrolyte to the anode, where they are stored. Upon discharge, these ions return to the cathode, releasing energy as they travel. This cyclical movement is what makes lithium batteries so effective.
However, this process is not without its challenges. Over time, the repeated movement of lithium ions can lead to electrode degradation. This degradation affects the battery's efficiency and lifespan. Understanding these limitations is crucial for improving future battery technologies. As research continues, we may find solutions to these issues, enhancing the reliability of lithium batteries even further.
This chart illustrates the relationship between the discharge rate (C-rating) and the capacity of a lithium battery. As the discharge rate increases, the capacity available decreases, showcasing how performance dynamics work in lithium batteries.
When exploring lithium battery performance, several key factors come into play. One crucial aspect is temperature. Lithium batteries operate optimally between 20°C and 25°C. Extreme temperatures can lead to decreased efficiency and lifespan issues. A study from the Journal of Power Sources indicates that high ambient temperatures can reduce battery capacity significantly by up to 20%.
Another important factor is charge cycles. The number of times a battery can be charged and discharged directly impacts its longevity. Data shows that a lithium battery typically endures about 500 to 1,500 cycles before capacity fades. Maintaining partial charges rather than full discharges can help extend battery life.
**Tips:**
- Store batteries in a cool, dry place.
- Avoid leaving them in high-temperature environments.
Additionally, chemical composition influences performance. Variations in electrolyte materials can enhance energy density or reduce safety risks. Understanding these differences is critical for manufacturers aiming to improve reliability.
**Tips:**
- Regularly monitor battery performance.
- Replace batteries when performance consistently deteriorates.
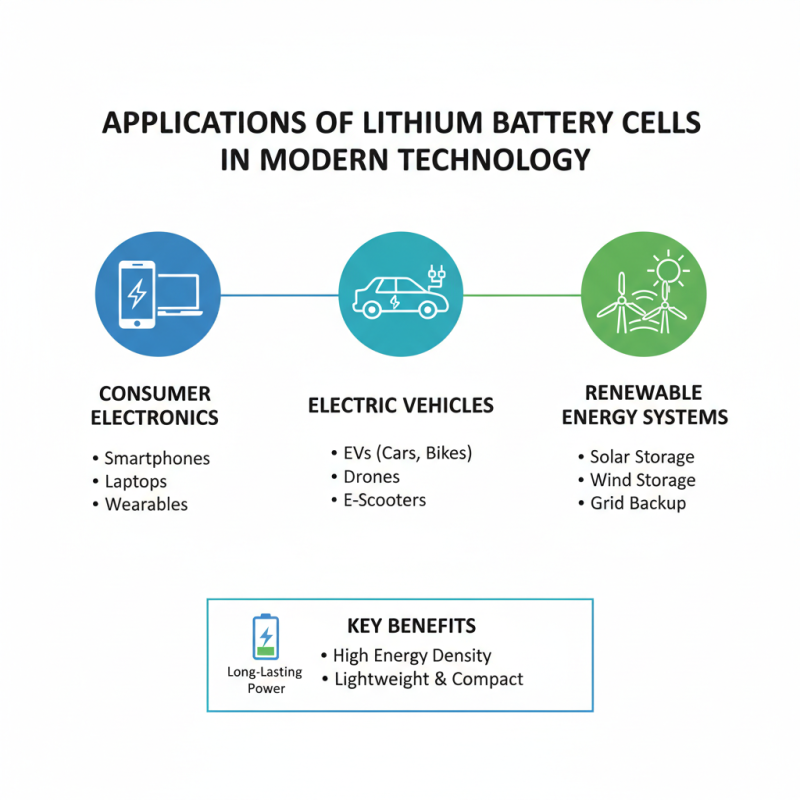
Lithium battery cells play a crucial role in modern technology. They are widely used in consumer electronics, electric vehicles, and renewable energy systems. These cells are lightweight and have a high energy density, making them ideal for devices that require long-lasting power. For example, smartphones and laptops rely on lithium batteries for extended usage without requiring frequent charging.
In addition to personal gadgets, lithium batteries are essential for electric vehicles (EVs). They power everything from compact scooters to large trucks. EV manufacturers focus on maximizing battery life and performance. The technology continues to evolve, with new materials enhancing energy storage capabilities. These advancements contribute to a cleaner environment by reducing dependency on fossil fuels.
However, challenges remain. Recycling lithium batteries is a pressing issue, as improper disposal can harm the environment. The supply chain for lithium extraction often faces scrutiny over ethical practices. Addressing these concerns is vital for the sustainable growth of battery technology. Constant research is necessary to improve battery life and sustainability, ensuring lithium batteries can meet future demands effectively.